
Allergen Testing in Food Products Explained: How ELISA Ensures Food Safety
Worried that a tiny trace of peanut, mustard, or egg in your snack could trigger an allergic reaction and curious how laboratories reliably find those hidden proteins? That concern is exactly why food allergen testing methods have become a critical part of modern food safety. Among them, the ELISA test for food allergens is one of the most widely used tools to detect even trace-level contamination before products reach consumers.
In this post, I’ll explain using India-focused context and clear numbers how the ELISA test for food allergens works, how sensitive today’s food allergen testing methods really are, and why those detection limits matter for both manufacturers and consumers. I’ll also connect the science to food allergen labeling regulations, showing what QA teams need to demand from labs to make label claims defensible and credible.
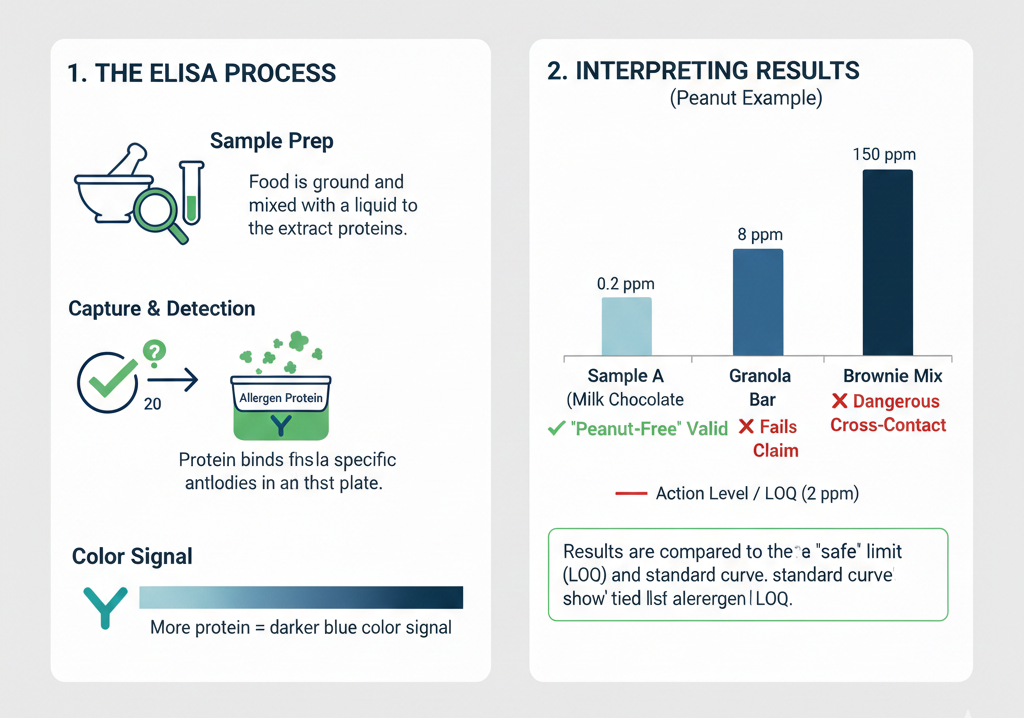
How ELISA finds hidden allergens and what the numbers mean
ELISA works by using antibodies that latch onto specific allergenic proteins (for example, peanut proteins such as Ara h components or egg ovomucoid). When the antibody binds, a visible reaction produces a numeric readout. That simplicity makes ELISA the go-to screening tool in many food labs.
Important, real numbers to remember:
- Typical limits of detection (LOD) for good commercial ELISA kits are in the range of 0.1–0.5 mg/kg (ppm) of allergenic protein in simple matrices.
- Practical limits of quantification (LOQ) labs report after validating in real foods commonly fall between 0.5–2 ppm, depending on how processed or complex the food is.
- Quality targets labs aim for on validated samples: recoveries of 70–120% and repeatability (RSD) under 20%.
Why this is useful: a kit claiming 0.1 ppm LOD on a clean test solution may behave differently in a baked cookie, a spicy mix, or a high-fat sauce. So the LOD/LOQ numbers are meaningful only when backed by matrix-specific validation, that is, tests done in the actual type of food after processing.
Why ELISA matters in India regulation, prevalence and real-world risk
India’s food labeling rules require honest ingredient lists for packaged foods, and manufacturers are increasingly expected to manage cross-contact risks and validate “free from” claims. The national regulator, Food Safety and Standards Authority of India, provides the legal framework that brands must follow, and enforcement is moving toward evidence-based testing.
On allergy prevalence: studies in India show mixed results because methods differ, but some child cohort studies report clinical peanut allergy figures as low as ~0.03%, while self-reported food allergy in adults can be up to ~3% in certain surveys. The key takeaway: even if population rates appear lower than some Western countries, sensitive individuals exist, and under-diagnosis is common so manufacturers cannot rely on low population prevalence as a reason to skip robust testing.
In practice this means: if you label a product “peanut-free” or “may contain nuts,” you should be able to show the testing data and validation that support that claim. ELISA remains the most practical routine tool to provide that evidence.
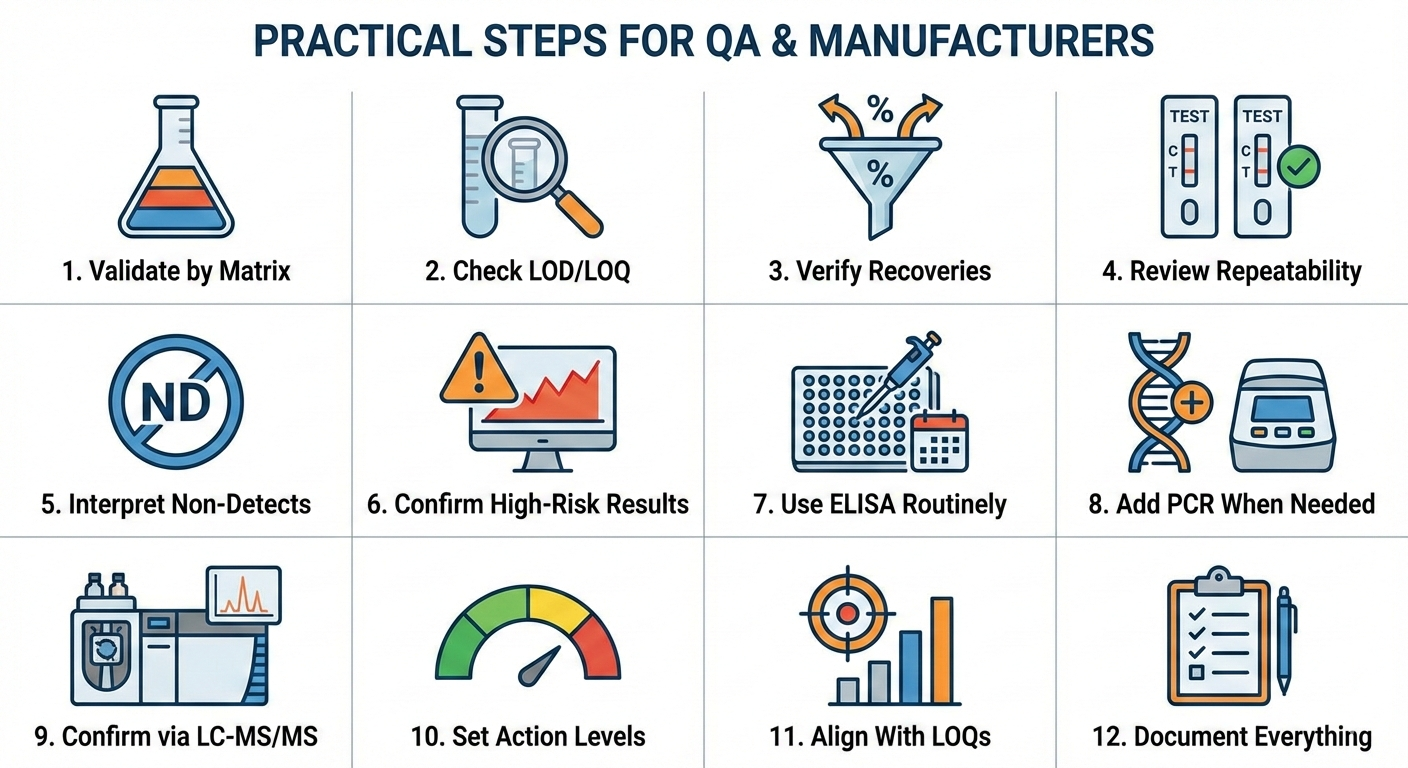
Practical steps QA teams and manufacturers should take
-
- Demand matrix-specific validation. Ask your lab for LOD, LOQ, recovery from incurred samples (spiked before processing), and repeatability figures. Don’t accept kit claims alone.
- Interpret non-detects correctly. A “no allergen detected” result only has meaning tied to the method’s LOQ in that specific food. For high-risk products, require orthogonal confirmation.
- Use complementary methods when needed. ELISA targets protein directly, ideal for most cases. But when protein is heavily processed or degraded (for example, high-temperature baked goods), labs can use PCR (DNA-based) as an indirect screen and LC-MS/MS (mass spectrometry) for peptide-level confirmation in regulatory or recall scenarios. A common workflow: ELISA for routine monitoring, LC-MS/MS for tough confirmations.
- Set defensible action levels. Rather than arbitrary zeros, set action levels based on validated LOQs and consumer risk (e.g., maintain control limits aligning with LOQs of 0.5–2 ppm for major allergens, adjusted by matrix).
5. Document everything. A defensible label claim includes the raw test data, validation tables, and the lab’s method statement showing sample prep and controls.
If you want testing and validation that includes matrix-specific ELISA, orthogonal confirmation options, and clear validation tables to support your labels, contact Anacon Laboratories they can help set up a defensible allergen testing program for your products.





